January 2026 marks a watershed moment for women’s health and molecular diagnostics in the United States. The Health Resources and Services Administration (HRSA) has officially updated its Women’s Preventive Services Guidelines to endorse human papillomavirus (HPV) self-collection as a primary screening option—a profound transformation in how the country approaches cervical cancer elimination, shifting the focus toward accessible, molecular-driven public health strategies.
Deep Dive: What the HRSA Guidelines Actually Say
The updated HRSA guidelines fundamentally shift the standard of care from traditional cytology (the Pap smear) to high-risk HPV (hrHPV) molecular testing. For women of average risk between the ages of 30 and 65, primary hrHPV testing every five years is now the preferred strategy. The most groundbreaking addition is that patient-collected hrHPV testing is officially designated as an appropriate method and must be offered as an option alongside clinician-collected testing. This validates molecular PCR technology as the definitive vanguard of early detection.
https://www.hrsa.gov/womens-guidelines
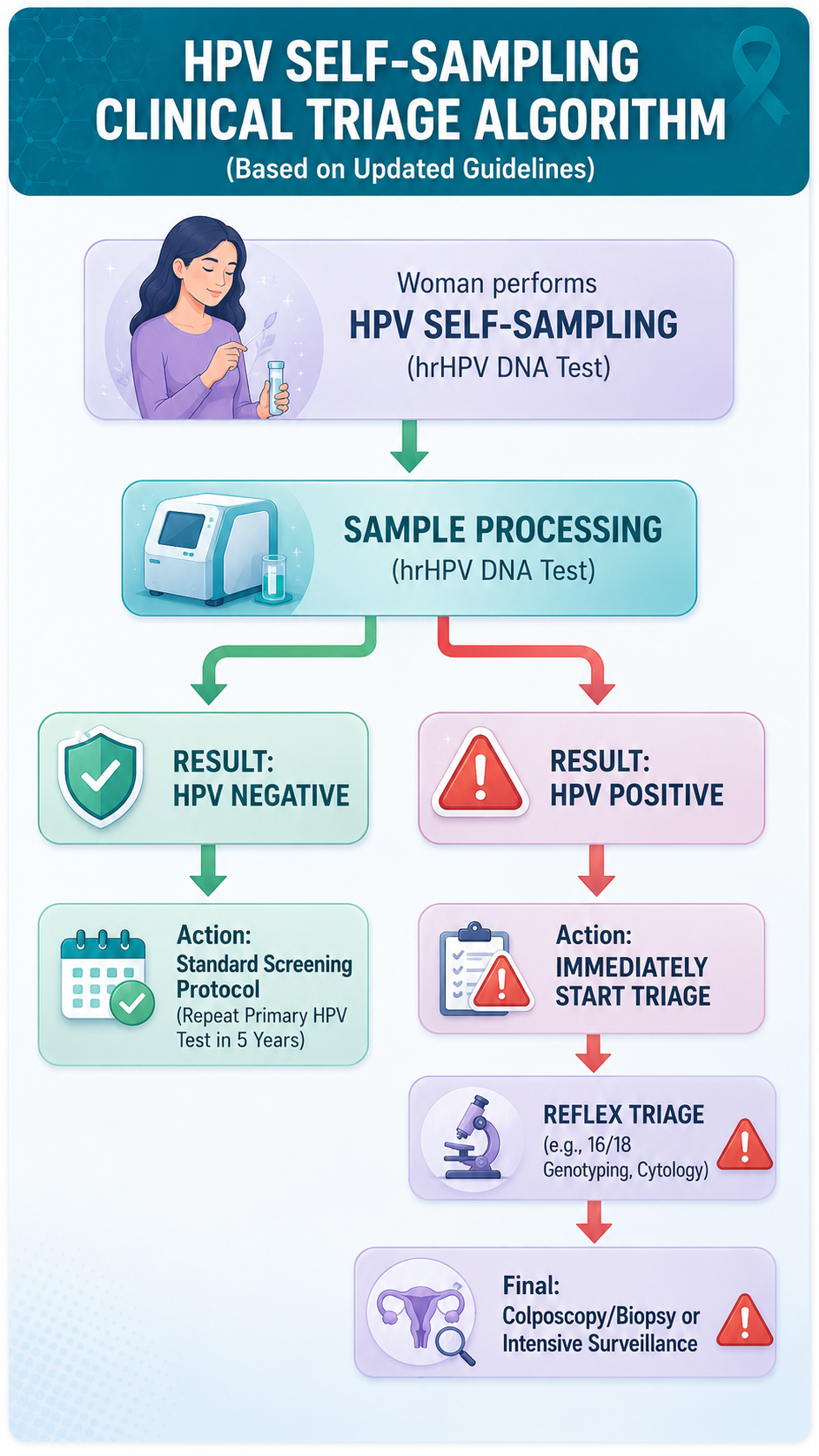
Steps and Crucial Precautions for Self-Sampling
While self-sampling democratizes access, the guidelines maintain strict clinical guardrails to ensure diagnostic integrity.
- The Procedure: Women can use an FDA-approved swab or brush to collect vaginal samples either in a clinical setting or, as the pathway clears, in the privacy of their homes.
- The Triage Step: Self-sampling is a screening tool, not a definitive diagnostic. If a patient tests positive for a high-risk HPV strain, the guidelines mandate immediate follow-up testing—such as cytology or colposcopy—to evaluate the presence of precancerous lesions.
- Precautions & Exclusions: The self-collection pathway is strictly for the “average-risk” population. It does not apply to women who are immunocompromised, living with HIV, exposed to diethylstilbestrol (DES) in utero, or those with a history of CIN2+ (cervical intraepithelial neoplasia grade 2 or higher) within the past 20 years. These individuals still require clinician-administered screening.
https://www.sgo.org/news/hrsa-announces-updated-cervical-cancer-screening-guidelines/
Why Is This a Monumental Milestone?
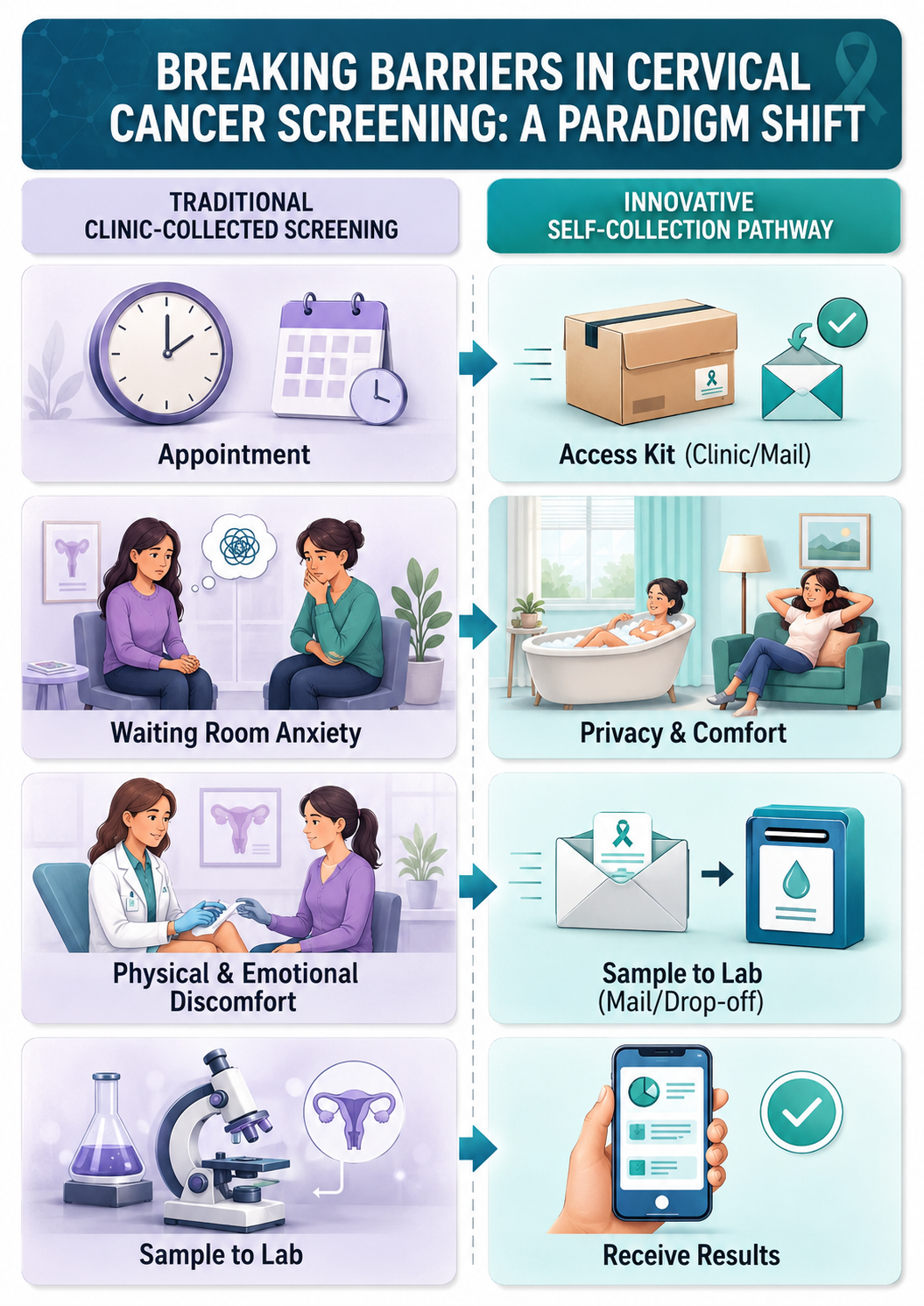
For decades, the speculum-based exam has been a significant barrier to care. Discomfort, cultural stigma, time constraints, and lack of healthcare access have led to alarming gaps—currently, 1 in 4 women in the U.S. are not up to date with their screening. Clinical trials such as the SELF-CERV study have demonstrated that self-swab tests are comparably accurate to clinician-collected samples, with 98% of users successfully collecting a valid sample, and 86% stating they would be more likely to stay up to date if they could test at home. Furthermore, the HRSA update requires private insurers to cover both the self-collection kits and any subsequent follow-up diagnostic testing without patient cost-sharing starting in 2027. Removing these financial and physical bottlenecks opens the door for massive, population-wide screening compliance.
The Global Landscape: A Worldwide Paradigm Shift
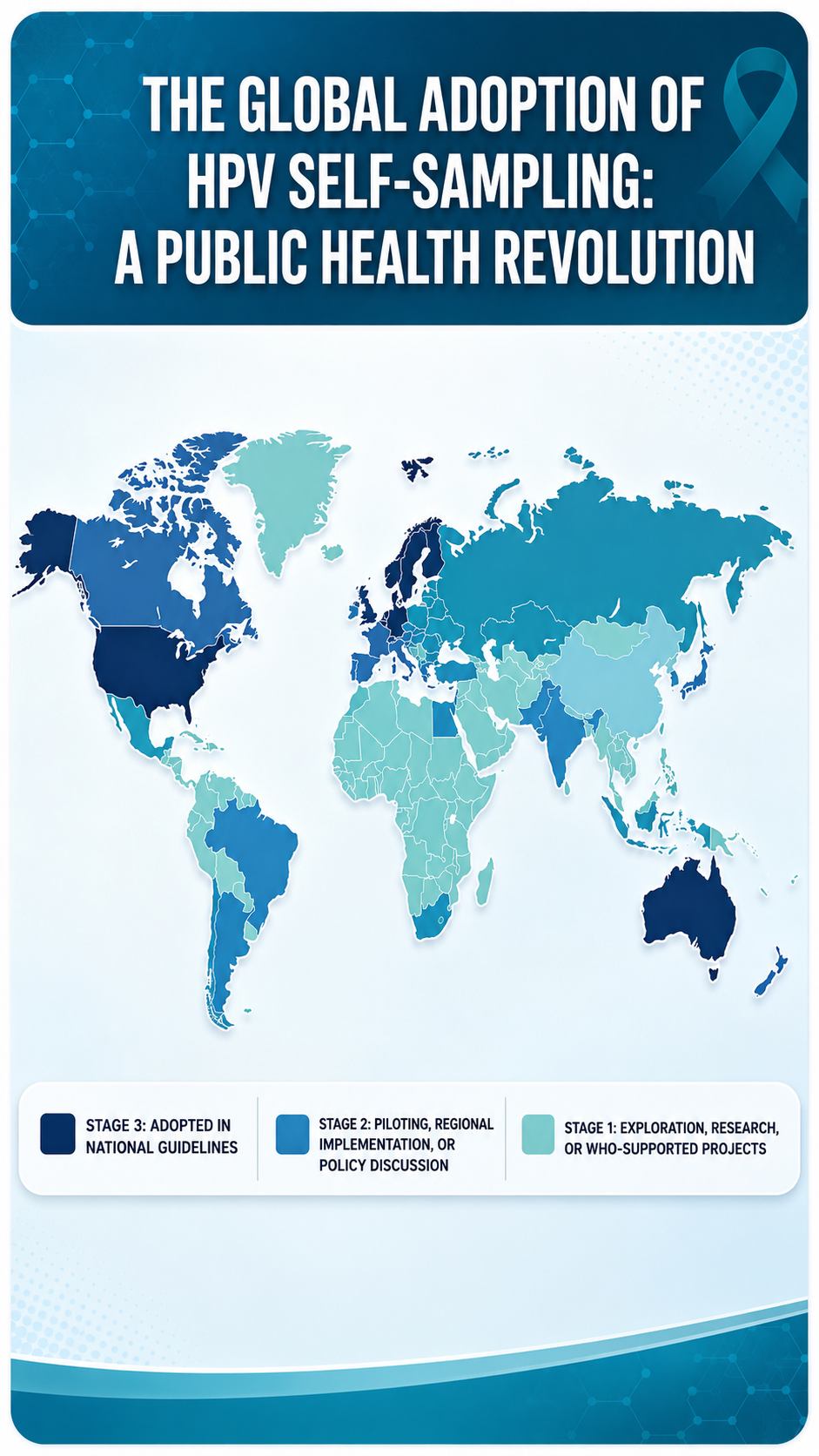
The U.S. is joining a well-established international movement toward self-sampling, providing excellent reference points for global market expansion.
- The Pioneers (The Netherlands & Australia): The Netherlands was the first country to implement a national mail-out program, sending self-sampling kits to all eligible women over 30. Australia allows women aged 25–74 to access self-collection kits through their healthcare providers, dramatically increasing engagement among under-screened communities.
- Europe’s Aggressive Push: Sweden deployed self-sampling kits to bypass pandemic lockdowns, sending them to 330,000 women in the Stockholm region. This single intervention boosted population test coverage from 75% to 85% in just one year, putting Sweden on an accelerated track to eliminate cervical cancer.
- Emerging Markets: Across Latin America (Argentina, Chile, Colombia) and various regions in Africa and Southeast Asia, national guidelines and WHO-backed pilot programs are increasingly leveraging self-sampling. This aligns directly with the WHO’s 2030 Cervical Cancer Elimination Initiative, which prioritizes accessible HPV DNA testing.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12677111/ ;
https://www.who.int/europe/news/item/08-09-2022-hpv-self-sampling-in-sweden-leading-to-faster-elimination-of-cervical-cancer
Looking Ahead: The IVD Industry’s Crucial Role
As self-sampling transitions from an alternative option to the global standard of care, the burden of performance shifts heavily to the laboratory infrastructure. High-throughput, automated PCR platforms capable of seamlessly processing diverse, self-collected sample types will be critical. The international market demands true “sample-in, result-out” efficiency to support these massive national screening rollouts. By breaking down the barriers of the clinic, molecular diagnostics can finally reach every corner of the globe.

