As the Southern Hemisphere transitions into autumn and winter, the 2026 respiratory virus season arrives carrying two parallel challenges.
The first is influenza — and the relentless pressure of viral evolution. The latest example is a new H3N2 branch researchers have labeled "subclade K," confirmed by Argentina's ANLIS National Institute through genomic sequencing in late 2025, with Brazil, Chile, and Bolivia now on heightened surveillance. This pattern of "a new variant arriving at season's end" is one the Southern Hemisphere has watched unfold for several years running.
The second comes from measles — a disease many had assumed was already defeated. In November 2025, the Pan American Health Organization (PAHO) officially announced that the Americas had lost their measles elimination status — a milestone the region first achieved in 2016, now gone. More worrying still: 2026 has opened worse than anyone predicted. In just the first three months, new measles cases across the Americas have already exceeded the total for all of 2025.
Two viruses, two trajectories — yet in hospital outpatient clinics, they look strikingly alike: fever, cough, malaise. What separates them? Increasingly, the answer is clear: the laboratory.
1. Two Pathogen Pressures Shaping the 2026 SH Season
1.1 Influenza: Variation Is the Norm, and South America Is Once Again on the Front Line
The 2025 SH influenza season followed a pattern that has become familiar: H1N1 led early on, with little drama in sight — until a new H3N2 branch, subclade K, surfaced in surveillance networks toward the season's end. This kind of mid-season shift plays out in different forms each year, and it is no stranger to South America: an early RSV surge in Brazil in 2023, an early influenza onset in Argentina in 2024, and now subclade K's late-season arrival in 2025.
The pattern is unmistakable: the Southern Hemisphere consistently stands at the front line of each new respiratory virus challenge.
Each new variant brings the same checklist for South American laboratories: identify the subtype quickly, report data to public health networks, and supply lead samples to reference labs. Early real-world evidence from England suggests that against the currently circulating H3N2 variants, vaccine effectiveness remains 72–75% in children and 32–39% in adults — still cutting hospitalization risk roughly in half, but no longer blocking infection as efficiently as in past years. Hospitals should plan for case counts higher than expected.
Adding to the complexity, subclade K rarely arrives alone. Australian data from 2025 showed that roughly 4.4% of patients tested positive for more than one respiratory virus simultaneously — influenza, RSV, SARS-CoV-2, hMPV. This kind of multi-pathogen co-circulation has shifted from exception to norm. WHO has accordingly updated the 2026 SH trivalent vaccine composition.
1.2 Measles: An Old Adversary Returns
If the influenza story is one of variation, the measles story is one of immunity gaps.
On November 10, 2025, PAHO announced that the Americas had lost their measles elimination status. The announcement was quiet, but its weight was enormous — the Americas had been the first, and remains the only, region in the world to have eliminated measles twice.
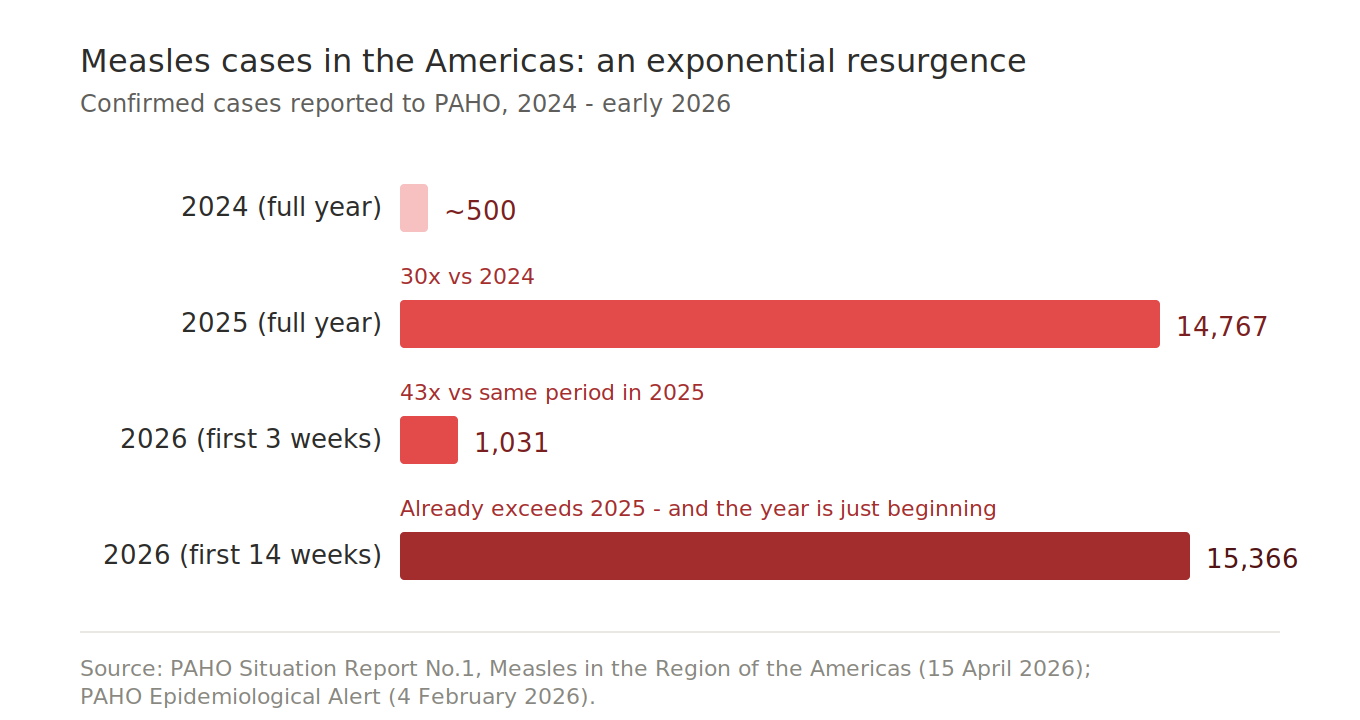
The numbers tell a stark story:
· 2024: ~500 measles cases across the Americas
· 2025: 14,767 cases — a 30-fold increase — with 32 deaths across 13 countries
· First 3 weeks of 2026: 1,031 cases — a 43-fold increase versus the same period in 2025
· First 14 weeks of 2026: 15,366 cases — already surpassing the entire 2025 total
In South America specifically, active outbreaks have been reported in Bolivia (597 cases), Brazil, Paraguay, Argentina, Uruguay, Chile, and Peru.
Why? The root cause is insufficient vaccine coverage. Only 33% of countries in the Americas have reached the 95% MMR first-dose coverage threshold — meaning most countries still hold sizeable pools of susceptible individuals. Once an imported case arrives, the virus finds a foothold.
And because MMR protects against both measles and rubella, coverage gaps mean rubella is also at risk — a quieter concern that may yet prove serious. For pregnant women, rubella infection can cause Congenital Rubella Syndrome (CRS), with newborns at risk of deafness, cardiac defects, and developmental delays. In its February 2026 alert, PAHO explicitly called for stronger laboratory diagnosis and outbreak response capacity.
2. Why "Detection" Has Become the Keyword for 2026
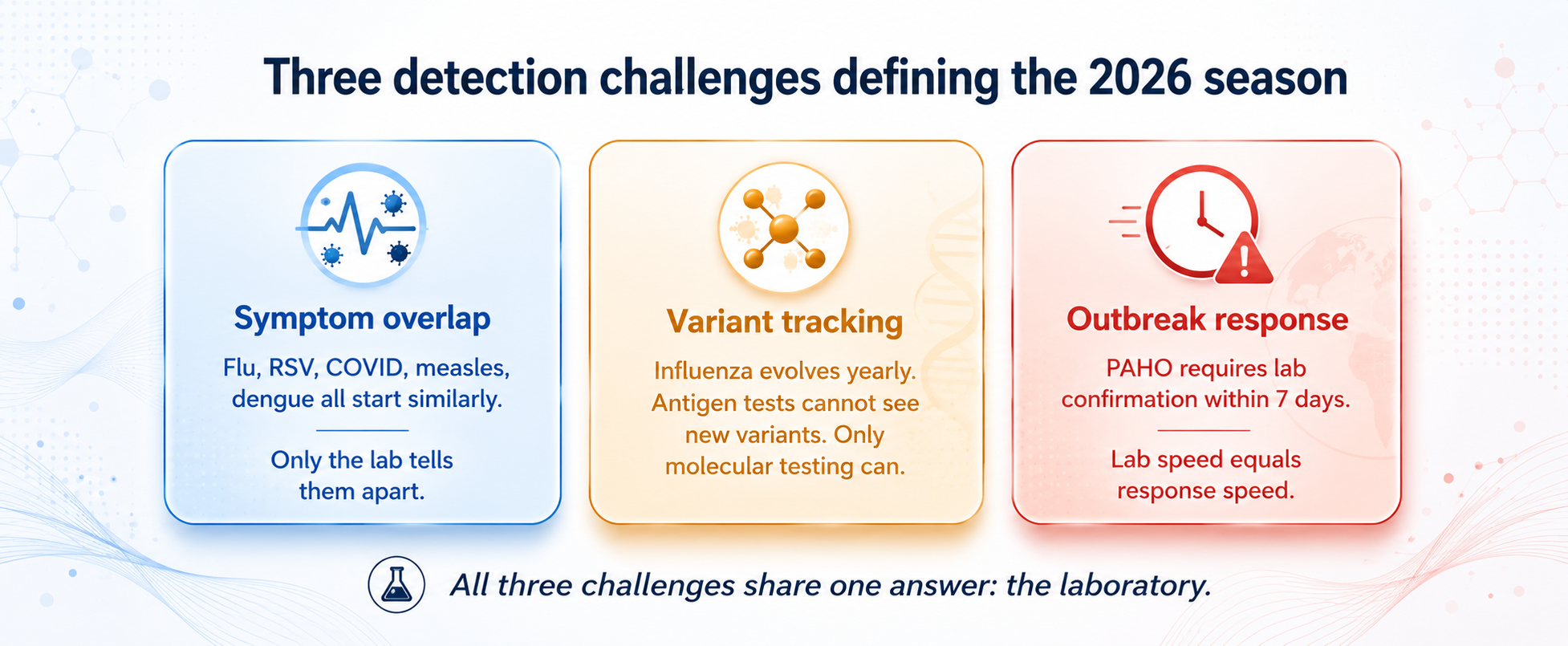
These two threat lines may look unrelated, but they put South American laboratories under the same spotlight.
Symptom overlap is the first detection challenge. A child with fever, cough, and a runny nose might have influenza, RSV, COVID-19, the early phase of measles — or even dengue, which is endemic across much of South America. Symptoms alone cannot tell a clinician whether to isolate, prescribe antivirals, or trigger an outbreak response. One accurate laboratory result is worth half a day of clinical guesswork.
Variant tracking is the second detection challenge. Influenza viruses evolve every year, with new branches and new code names appearing constantly. Antigen rapid tests cannot see these changes — only molecular testing can, and ideally a PCR assay capable of distinguishing H1 from H3 subtypes, providing early signals of variant activity.
Outbreak response is the third detection challenge. PAHO requires laboratory confirmation of suspected measles cases within seven days. Every day of delay means another circle of potential transmission. Laboratory speed equals public health response speed.
3. Influenza Detection: Three Capability Priorities for 2026
3.1 Multi-Pathogen Detection in a Single Reaction
The integrated surveillance algorithm PAHO has rolled out across the Americas centers on influenza + SARS-CoV-2 multiplex PCR as the standard. At the clinical laboratory level, adding RSV to form a four-target panel has become an increasingly common configuration — better suited to handle the diagnostic complexity of RSV and influenza co-circulation in pediatric and elderly populations. More advanced panels extend coverage to hMPV, parainfluenza, adenovirus, rhinovirus, and other targets in 7+ pathogen panels — particularly valuable for pediatric ICU and severe respiratory infection cases. One sample, one reaction, multiple answers — significantly shortening turnaround time and accelerating triage, isolation, and antiviral or monoclonal antibody decisions.
3.2 Influenza Subtyping (H1/H3): The Bridge Between Clinic and Surveillance
Influenza viruses evolve every year, and new branches and new code names keep coming — this is a reality frontline laboratories must accept by default. A PCR assay capable of distinguishing H1 from H3 lets a laboratory answer not just "is it influenza?" but also "which subtype is it?" — and that is the entry ticket into global surveillance networks like the PAHO Respiratory Viruses Dashboard and WHO FluNet.
The international standard is a "two-step" workflow: frontline laboratories use PCR to identify H3-positive samples, then forward them to next-generation sequencing (NGS) to confirm the specific subclade. This is the workflow PAHO, the UK Health Security Agency (UKHSA), and Canada's National Microbiology Laboratory all use to track new influenza variants — and subtyping PCR sits at the very front of that surveillance chain.
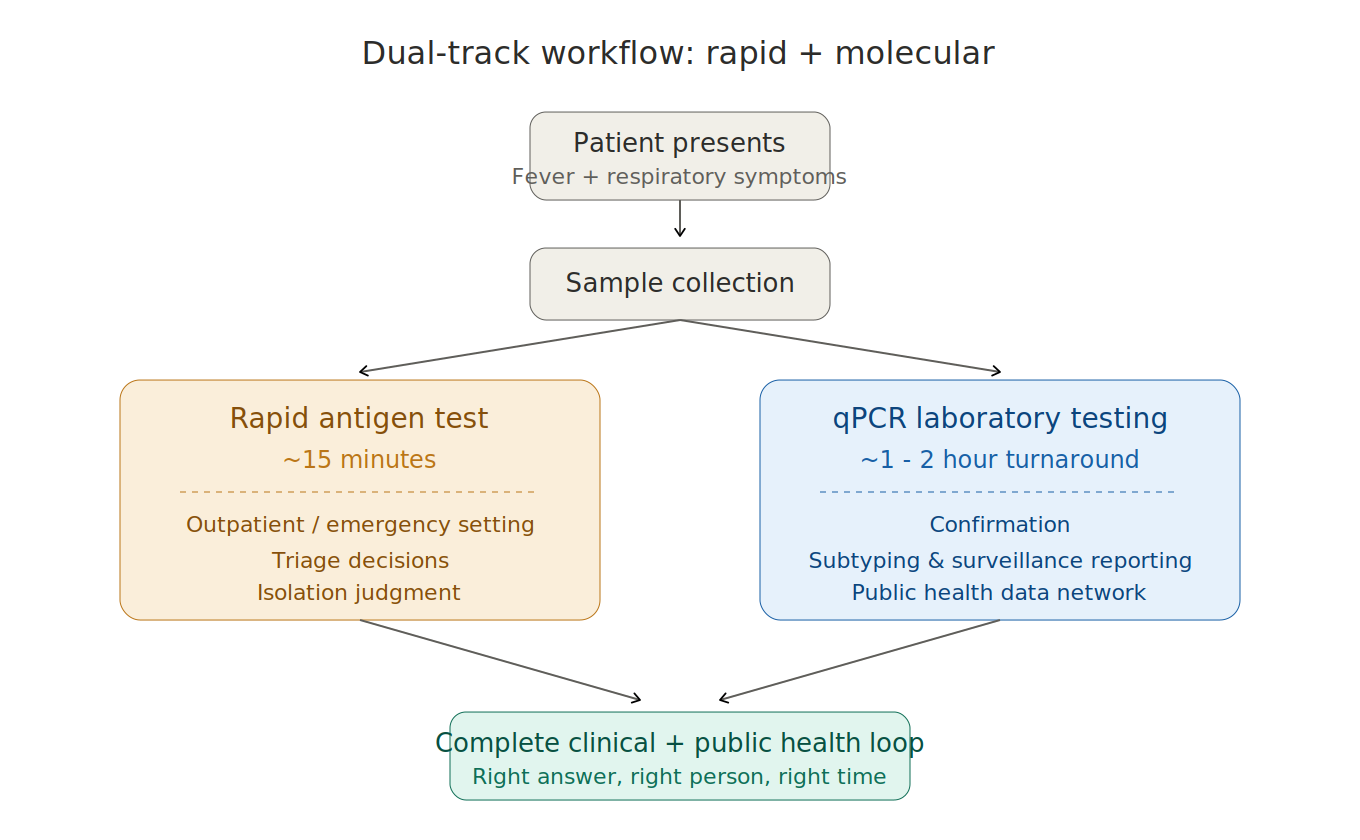
3.3 A Dual-Track Workflow: Rapid + Molecular
For outpatient and emergency settings, antigen rapid tests (~15 minutes) support frontline triage and isolation decisions for high-risk patients. For laboratory settings, qPCR (~1–2 hour turnaround) provides confirmation, subtyping, and surveillance reporting. The two are complementary, not interchangeable — the first answers "should this patient be isolated now?", the second answers "what is the pathogen, and is it changing?" In the 2025 SH season, this dual capability proved crucial during the early RSV surges in Argentina and Brazil.
4. Measles & Rubella Detection: Three Priorities for 2026
4.1 PCR Has Become the Surveillance Gold Standard
Traditional serology has three limitations in measles surveillance: a late detection window (IgM stabilizes only 3–5 days after rash onset), cross-reactivity with antibodies to other viruses such as dengue, and false positives in those recently vaccinated with MMR. PCR detects viral RNA in the early rash phase — and even during the prodrome — with high negative predictive value, and supports genotyping to distinguish imported from endemic transmission. This is the core evidence base PAHO uses through its integrated surveillance information system to assess elimination status.

4.2 Differential Diagnosis Across the Fever-Rash Spectrum
Measles and rubella can clinically resemble dengue, Zika, parvovirus B19, and drug rashes — particularly in tropical and subtropical South America, where these conditions co-circulate. Multiplex PCR for measles and rubella distinguishes both pathogens in a single reaction. This matters most for pregnant women, where missed rubella diagnosis can lead to Congenital Rubella Syndrome (CRS) — a lifelong consequence avoidable through timely diagnosis.
4.3 Outbreak Response Speed Is Limited by Laboratory Capacity
PAHO recommends a maximum 7-day turnaround from sample collection to result for suspected cases — yet laboratory confirmation is currently a bottleneck in many regions. Distributed qPCR capacity — including automated nucleic acid extraction, reagent stability and cold-chain adaptability, and localized SOPs and training — represents critical infrastructure investment for measles and rubella response in the 2026 SH season. The data interface with public health networks matters equally: laboratory results that fail to flow into PAHO and national surveillance systems lose much of their value.
Closing: The Earlier the Preparation, the Steadier the Response
The defining question for the 2026 SH respiratory season is not "how quickly can we react?" but "how deep have we prepared?"
The continuing evolution of influenza and the resurgence of measles point to the same answer: detection capacity is the rate-limiting step in public health response. Multi-pathogen panels, influenza subtyping, molecular differentiation of measles and rubella, and locally deployable PCR — these four capabilities together form the strategic core for South American laboratories in the 2026 season.
Respiratory virus seasons have never rewarded a "wait and see" approach. The 2026 season is already at the door. The real question is not whether it is coming — but whether the laboratory will be ready when it does.
About BioPerfectus
For South America's laboratories and public health programs facing the 2026 SH respiratory season, what is needed is detection capacity that can simultaneously address a shifting influenza landscape and a measles resurgence.
BioPerfectus provides comprehensive infectious disease diagnostic solutions to laboratories in over 100 countries and regions, covering influenza, RSV, SARS-CoV-2, measles, rubella, and other respiratory and fever-rash syndrome testing needs. Our portfolio includes:
· Rapid antigen tests — Influenza A & B Rapid Test Kit, for frontline triage
· Multiplex Real-Time PCR kits — Allfastra SARS-CoV-2 / Flu A / Flu B / RSV (4-target) and Respiratory Virus Panel (7-target)
· Influenza A and B PCR detection — Influenza A and B Viruses Real Time PCR Kit
· Measles and rubella detection — Measles and Rubella Viruses Real-Time PCR Kit
· Integrated automation platform — SAW automated workstations + STC qPCR systems
To explore our respiratory and measles/rubella diagnostic portfolio: https://www.bioperfectus.com/productonelist?C=101
References
1. PAHO. Briefing note: Influenza A(H3N2) subclade K (J.2.4.1), considerations for the Americas Region. 11 December 2025. https://www.paho.org/en/documents/topics/influenza-sars-cov-2-rsv-and-other-respiratory-viruses
2. PAHO. Epidemiological Alert: Simultaneous circulation of seasonal influenza and respiratory syncytial virus in the Americas. 9 January 2026. https://www.paho.org/en/news/10-1-2026-paho-issues-alert-simultaneous-circulation-seasonal-influenza-and-respiratory
3. PAHO. Epidemiological Alert: Measles in the Americas Region. 4 February 2026. https://www.paho.org/sites/default/files/2026/02/2026-feb-3-phe-alerta-epi-measles-enfinal_0.pdf
4. PAHO. Situation Report No.1: Measles in the Region of the Americas. 15 April 2026. https://www.paho.org/en/documents/situation-report-no1-measles-region-americas-15-april-2026
5. PAHO. PAHO calls for regional action as the Americas lose measles elimination status. 10 November 2025. https://www.paho.org/en/news/10-11-2025-paho-calls-regional-action-americas-lose-measles-elimination-status
6. PAHO. Influenza and SARS-CoV-2 integrated surveillance laboratory testing algorithm. October 2025. https://www.paho.org/en/documents/influenza-and-sars-cov-2-integrated-surveillance-laboratory-testing-algorithm
7. WHO. Recommended composition of influenza virus vaccines for use in the 2026 southern hemisphere influenza season. September 2025. https://www.who.int/publications/m/item/recommended-composition-of-influenza-virus-vaccines-for-use-in-the-2026-southern-hemisphere-influenza-season
8. Separovic L, Sabaiduc S, Zhan Y, et al. Interim 2025/26 influenza vaccine effectiveness estimates with immuno-epidemiological considerations for A(H3N2) subclade K protection, Canada, January 2026. Eurosurveillance. 2026;31(5):2600068. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12881843/
9. UK Health Security Agency / Eurosurveillance. Early influenza virus characterisation and vaccine effectiveness in England in autumn 2025, a period dominated by influenza A(H3N2) subclade K. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12639273/
10. MercoPress. Superflu makes it to Argentina and Brazil. 19 December 2025. https://en.mercopress.com/2025/12/19/superflu-makes-it-to-argentina-and-brazil
11. Australian Centre for Disease Control. Australian Respiratory Surveillance Reports – 2025. https://www.cdc.gov.au/resources/collections/arsr-2025
12. PAHO Respiratory Viruses Surveillance Dashboard. https://dashboards.pahoflu.com/app/respiratory_viruses/

